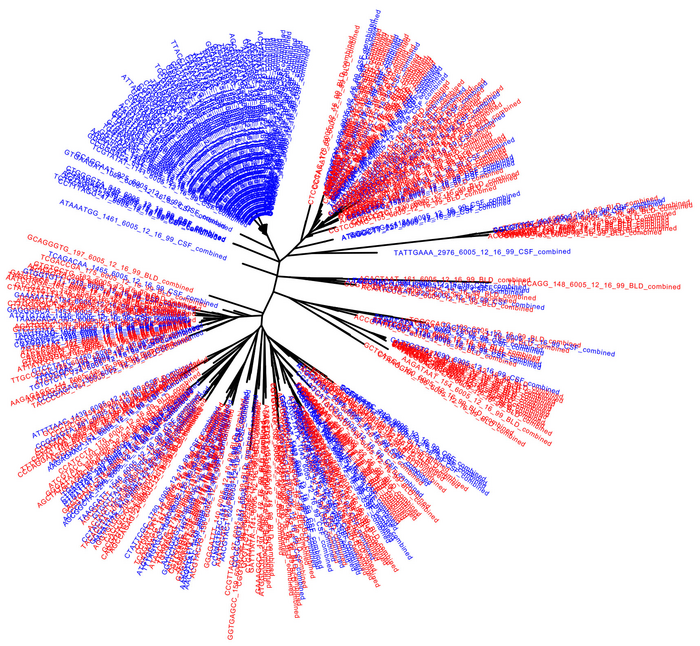
Rebound DNA sequences from the blood (red) and the CSF (blue) [Swanstrom Lab, UNC School of Medicine]
HIV’s ability to lay dormant, during suppressive antiviral therapy, is a major barrier to a cure. The latent reservoir in infected memory CD4+ T cells (in lymphoid tissue) is the best characterized. However, once antiretroviral therapy (ART) is stopped, “rebound” virus becomes detectable—HIV that re-emerges with new cells getting infected. This problem of latency remains a major goal for researchers trying to create curative therapies for HIV.
Now, scientists describe another layer to the challenge of HIV latency. Ron Swanstrom, PhD, director of the UNC Center for AIDS Research, and colleagues analyzed rebound virus in the cerebral spinal fluid (CSF) during the period when people had just stopped taking ART. Their findings provide indirect evidence for the existence of a distinct latent reservoir of CD4+ T cells in the central nervous system (CNS).
This work is published in Nature Microbiology in the paper, “Rebound virus in the cerebrospinal fluid reveals a possible HIV-1 reservoir.”
“Our analysis of rebound virus suggests latently infected T cells in the CNS are separate from the latent reservoir in the blood,” said Swanstrom. “Our analysis allows us to infer the presence of a distinct pool of latently infected cells in the CNS waiting to reinitiate infection once ART is interrupted.”
The researchers compared the genetic sequences of rebound virus particles when ART was discontinued in 11 human participants. This approach allowed the scientists to assess the similarities between viral populations in the blood and CSF to determine whether they were part of a common latent reservoir. In many cases, the viral populations were not the same, which suggested they can represent different populations of latently infected cells.
The researchers also studied details of viral replication to determine if rebound virus had been selected for replication in CD4+ T cells—the primary home of the virus—or had evolved to replicate in central nervous system myeloid cells, such as macrophages and microglia. All rebound viruses tested were adapted to growth in T cells. For several participants, the researchers also compared viral populations in blood and CSF before ART initiation and after ART was stopped.
These experiments provide further evidence that HIV-infected CD4+ T cells can cross over from blood into the CNS, but also that some latently infected cells may be resident in the CNS during therapy. Any curative therapy would need to activate this dormant reservoir, as well as the latent reservoir in the blood and lymph tissue.

