With SARS-CoV-2’s delta variant raising concerns, and COVID-19 case counts rising, the need for quick, cheap, diagnostic tests has not diminished over the course of the pandemic. Improvements on the RT-PCR test, the current standard of COVID-19 testing, could enhance how public health measures are implemented for the duration of the COVID-19 pandemic, and future pandemics to follow.
Now, engineers at MIT and Harvard University have designed a test that can detect SARS-CoV-2 from a saliva sample in about an hour with the same accuracy as the PCR tests now used. The small, tabletop device can also be used to detect specific viral mutations linked to some of the SARS-CoV-2 variants that are now circulating. Obtaining results in an hour could potentially make it much easier to track different variants of the virus, especially in regions that don’t have access to genetic sequencing facilities.
The new diagnostic is described in Science Advances in the paper, “Minimally instrumented SHERLOCK (miSHERLOCK) for CRISPR-based point-of-care diagnosis of SARS-CoV-2 and emerging variants.”
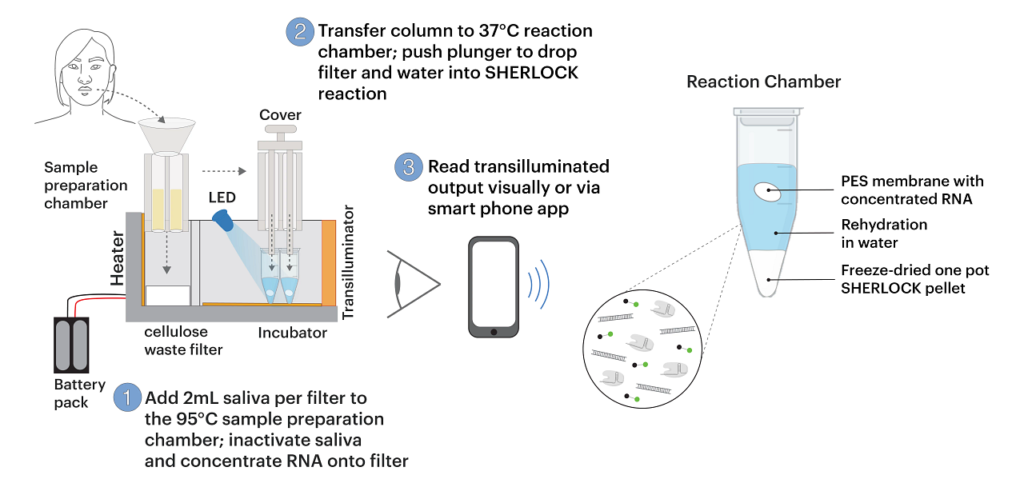
“Our goal was to create an entirely self-contained diagnostic that requires no other equipment,” said Xiao Tan, MD, PhD, a clinical fellow at the Wyss Institute and an instructor in gastroenterology at Massachusetts General Hospital. “Essentially the patient spits into this device, and then you push down a plunger and you get an answer an hour later.”
The new diagnostic is based on SHERLOCK, a CRISPR-based tool that James Collins, PhD, professor of medical engineering and science in MIT’s Institute for Medical Engineering and Science (IMES) and the department of biological engineering, and others first reported in 2017. Components of the system include an RNA guide strand that allows detection of specific target RNA sequences, and Cas enzymes that cleave those sequences and produce a fluorescent signal. All of these molecular components can be freeze-dried for long-term storage and reactivated upon exposure to water.
Last year, Collins’ lab began working on adapting this technology to detect the SARS-CoV-2 virus, hoping that they could design a diagnostic device that could yield rapid results and be operated with little or no expertise. They also wanted it to work with saliva samples, making it even easier for users. To achieve that, the researchers had to incorporate a step that disables salivary nucleases which destroy nucleic acids such as RNA. Once the sample goes into the device, the nucleases are inactivated by heat and two chemical reagents. Then, viral RNA is extracted and concentrated by passing the saliva through a membrane.
“That membrane was key to collecting the nucleic acids and concentrating them so that we can get the sensitivity that we are showing with this diagnostic,” noted Rose Lee, MD, an instructor in pediatrics at Boston Children’s Hospital and Beth Israel Deaconess Medical Center and a visiting fellow at the Wyss Institute.
This RNA sample is then exposed to freeze-dried CRISPR/Cas components, which are activated by automated puncturing of sealed water packets within the device. The one-pot reaction amplifies the RNA sample and then detects the target RNA sequence, if present.

“We demonstrated that our platform can be programmed to detect new variants that emerge, and that we could repurpose it quite quickly,” said Collins. “In this study, we targeted the U.K., South African, and Brazilian variants, but you could readily adapt the diagnostic platform to address the Delta variant and other ones that are emerging.”

“The ability to detect and track these variants,” said Helena de Puig, PhD, postdoc at Harvard University’s Wyss Institute for Biologically Inspired Engineering and first author on the paper, “is essential to effective public health, but unfortunately, variants are currently diagnosed only by nucleic acid sequencing at specialized epidemiological centers that are scarce even in resource-rich nations.”