
More than a decade ago, the words of the late Christopher Reeve inspired Grégoire Courtine, Ph.D., associate professor at the Swiss Federal Institute of Technology Lausanne (EPFL) to focus his research in the area of spinal cord injury with the singular aim to make patients’ lives better. Today, the report from his lab that three patients with chronic paraplegia have regained the ability to walk through targeted neurotechnology shows that he is well on his way to achieving that goal.
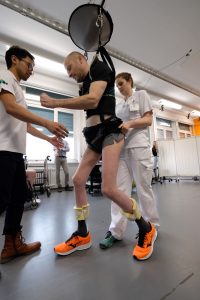
All three participants in the STIMO (STImulation Movement Overground) study, who had sustained cervical spinal cord injuries many years ago, are now able to walk with the aid of crutches or a walker.

The work is published in two papers, out today, that report on different aspects of the project.
One paper published in Nature, titled, “Targeted neurotechnology restores walking in humans with spinal cord injury,” describes how the Swiss team introduced targeted spinal cord stimulation neurotechnologies that enabled voluntary control of walking in individuals who had sustained a spinal cord injury more than four years ago.
How did they do it? The authors explain that they used an implanted pulse generator with real-time triggering capabilities to deliver trains of spatially selective stimulation to the lumbosacral spinal cord with timing that coincided with the intended movement.
The participants were surgically implanted with an array of 16 electrodes over the lumbar region of the spinal cord, outside of the protective dura mater. This electrode array is connected to an implantable pulse generator which can be controlled with a personalized voice-controlled watch to administer short bursts of electricity. Because different areas of the spinal cord are activated during the different movements associated with walking, this system allows for preferential activation of those areas when the participant is trying to walk.
“All the patients could walk using body weight support within one week. I knew immediately that we were on the right path,” recalls Jocelyne Bloch, M.D., neurosurgeon at Lausanne University Hospital (CHUV) who surgically placed the implants in the patients. Indeed, within one week, this spatiotemporal stimulation had re-established adaptive control of paralyzed muscles during overground walking, and locomotor performance improved during rehabilitation.
“One of the most exciting pieces of this work,” according to Peter Grahn, Ph.D., senior engineer at the Mayo Clinic Neuroengineering Laboratory, is “the observation of sustained functional gains when stimulation was turned off.” After a few months, participants regained voluntary control over previously paralyzed muscles without stimulation and could walk or cycle in ecological settings during spatiotemporal stimulation.
This is not the first time that this type of research has allowed previously paralyzed people to walk. Indeed, just over a month ago, two papers made a big splash in the same field: one from the Mayo clinic published in Nature Medicine and from the Harkema lab at the Kentucky Spinal Cord Injury Research Center at the University of Louisville, published in NEJM.
So, what makes this paper different? Edelle Field-Fote, Ph.D., director of the Spinal Injury Research & The Hulse Spinal Injury Laboratory and professor at Emory University School of Medicine, explains that “the difference in this approach is that previous groups stimulated everything in the area.” She says that it is like cooking a pot of beans in a pot. “What other groups have done is to turn on all of the burners on the stove to heat one pot of beans. But Dr. Courtine’s group has found a way to turn on just the burner that is needed. In targeting the stimulation to only the important areas, his group has steered the activation that is coming down from the brain.”
The companion paper published in Nature Neuroscience is entitled “Electrical spinal cord stimulation must preserve proprioception to enable locomotion in humans with spinal cord injury.” In this paper, the authors hypothesized that part of the reason that EES has had limitations to date is due to an interference between EES and proprioception, or the ability to know where your limbs (or other body parts) are in space. The results suggest that EES blocks a significant amount of proprioceptive input in humans, but not in rats.
Dr. Field-Fote explains that this finding makes Dr. Courtine’s targeted approach of stimulation much more striking. In fact, the previous approaches of more general stimulation, where the proprioception may have been blocked, could have created an obstacle that patients needed to overcome. This may also explain why previous methods take longer training regimens to see improvement. Having this information has allowed Dr. Courtine’s team to identify stimulation parameters that don’t block the proprioceptive information.
Dr. Field-Fote says that she “tends to be rather critical, but this group did a really good job” and that the work has “a lot of value for real-world function, mainly because it doesn’t require months of training.” It also expands the evidence to those with different spinal cord severity classifications which, to our knowledge, has not been studied before, according to Kristin Zhao, Ph.D., director of the Mayo Clinic Assistive and Rehabilitative Technologies Laboratory.
Dr. Courtine refers to this as a “proof of principle” and is mindful to set realistic expectations for spinal cord patients. Dr. Zhao tells GEN that “the application of epidural electrical stimulation to enable functions lost due to spinal cord injury is in its early stages,” adding that the next steps “will involve improving our understanding of the relationship between physical therapy, stimulation parameters, and spinal network plasticity over long-term stimulation and addressing which patients will respond successfully to the intervention.” Dr. Courtine’s goal is to start the process in patients earlier, immediately after the injury.
“While these results are exciting, several hurdles remain before widespread translation of this technology can become a reality,” says Dr. Grahn. “For example, before regulatory approval can be achieved for widespread use in humans with spinal cord injury, injury profiles that respond to spinal neuromodulation must be clearly defined.”
Dr. Grahn states that “as research continues to improve technology and scientific knowledge of spinal neuromodulation after paralysis, the potential is great to translate these discoveries into feasible therapeutics.” Dr. Zhao adds that “the field is moving forward and this is an exciting time that holds much promise for individuals with spinal cord injury.” However, calibrating expectations is important and Dr. Zhao adds the important note that “these methods are only available as part of research protocols and not as a clinical treatment.”
Dalton Dietrich, Ph.D., scientific director of The Miami Project to Cure Paralysis and professor at the University of Miami, and deputy editor of the Journal of Neurotrauma (A Mary Ann Liebert Journal) is hopeful, telling GEN, “Together these targeted EES technologies offer real hope for improving clinically relevant function in people living with severe or complete paralysis.”
There is reason for Dr. Dietrich’s optimism. Dr. Courtine has made huge strides in a small amount of time. In his 2013 TED talk, “The paralyzed rat that walked,” Dr. Courtine explains that the neural network necessary and sufficient to coordinate locomotion is present below most injured spinal cords. But, they are dormant because the input from the brain is interrupted. His idea, all along, has been to awaken this network and he proved that he could do it in rats. Now, just five years later, he is able to do it in humans.



