January 15, 2011 (Vol. 31, No. 2)
Case Study Demonstrates Utility of Inventive Strategies
Enveloped viruses have tremendous potential in a number of areas of great interest to biotech and pharma firms including gene therapy and vaccination. These viruses, which are typically produced in animal cell systems, are expensive to manufacture. As a result, identification of biological and engineering strategies that are scalable and cost-effective is paramount.
The complexity of these biological products, however, has hampered the development of robust downstream processes. Obtaining the quantities required for preclinical and clinical trials is especially problematic.The labile lipid membrane layer that harbors glycoproteins (often critical for infection) over the viral capsid further increases the challenges inherent in the processing of enveloped viruses.
Scientists at the Animal Cell Technology Unit of iBET are using recombinant baculoviruses as an enveloped virus model in order to optimize this task. Recombinant baculoviruses are widely used as vectors for the production of recombinant proteins in insect cells.
More recently, these viruses have been gaining attention due to their emerging potential as gene-therapy vehicles. While their production in stirred bioreactors using insect cells is an established technology, downstream processing of baculoviruses intended for clinical applications is only now catching up.
This article will discuss the evaluation of a scalable, cost-effective downstream processing strategy based on membrane processes. The evaluated process comprised three steps—depth filtration, ultra/diafiltration, and membrane sorption (Figure 1). Global recovery yields of clinical-grade material reached 40% using easy-to-scale-up technologies under cGMP guidelines. This constituted a major advance over the much lower yield and nonscalable purification process based on ultracentrifugation density gradients (Figure 1).
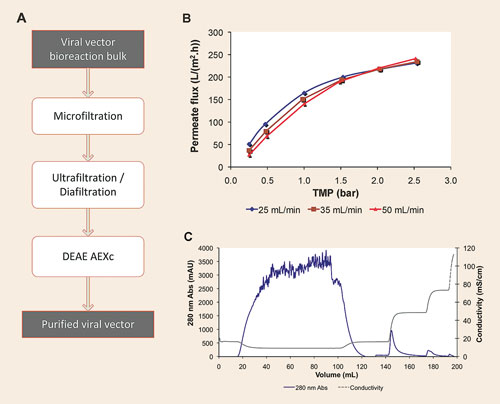
Figure 1. (A) Downstream process flow diagram; (B) Selection of optimal TMP and retentate flow rates at the tangential flow filtration step; (C) Sartobind D run of a bind-elute anion-exchange chromatography: baculoviruses are collected in the first elution peak. [B and C reproduced from Gene Therapy, 16:766-775 with permission of Nature Publishing Group.]
Methods
Sf9 insect cells were grown in Sf900-II serum-free medium (Life Technologies) to produce recombinant baculoviruses. The cells were infected at a multiplicity of infection of 0.1 infective viruses per viable cell. The bioreaction was carried out using a disposable Wavebag bioreactor with a 25 L working volume. When the cell viability was lowered to about 50%, the bulk was harvested and submitted to clarification.
Microfiltration was integrated downstream of the bioreactor via a series of two depth filters—3 µm and 0.65 µm (Sartorius Stedim Biotech). This strategy prevented baculoviruses from becoming entrapped within the pores (these rod-shaped viruses are up to 400 nm long). Tangential flow ultrafiltration and diafiltration were performed using 100 kDa Hydrosart cassettes (Sartorius Stedim Biotech). An ÄKTAcrossflow (GE Healthcare) system was used to operate each filtration step autonomously. The bulk was concentrated sixfold and diafiltered with two diafiltration volumes continuously.
Anion-exchange membrane chromatography was used as a capture step utilizing Sartobind D membrane adsorber units (Sartorius Stedim Biotech). Elution profiles were generated with increasing NaCl concentrations up to 1.5 M NaCl on PBS equilibration buffer supplemented with Ca2+ and Mg2+. An ÄKTAexplorer 100 system (GE Healthcare) was used to perform these runs.
Virus genomes were quantitated using real-time PCR (Roche Diagnostics) tracing a specific baculovirus gene and by TCID50 end-point dilution assay using Sf9 cells (infective titer).
Zeta-potential was measured by dynamic light scattering (DLS) using Zetasizer NanoZS (Malvern Instruments) in low conductivity phosphate buffers; hydrodynamic size assessment was carried out using the same equipment. Surface plasmon resonance (SPR) experiments were performed with the Biacore 2000 system (GE Healthcare) at 25ºC, using 20 mM phosphate buffers with the specified NaCl supplementation and at a constant pH 6.8. Sensors chips were derivatized with diethylaminoethyl weak anion-exchanger ligand.
Results
Based on purification steps that are easily scalable, a downstream processing workflow was established. The use of membrane processes facilitated the use of disposable formats, which resulted in cleaning and validation cost savings.
A series of two depth filters integrated downstream of the bioreactor allowed for efficient passage of the recombinant baculoviruses while efficiently removing cells and cell debris. Tangential flow filtration of up to sixfold concentration with a subsequent diafiltration step of two volumes (Figure 1B, Table) was achieved, allowing concentration and primary purification of LMW impurities for shorter times as compared to ultracentrifugation.
Use of the Sartobind D membrane adsorber as an anion-exchange chromatography step constituted a fast purification protocol with decreased buffer consumption and recovery yields up to 65% (Table).
To improve performance of the anion-exchange chromatography step, SPR and DLS scale-down methodologies were developed to increase the fundamental understanding of this unit operation.

Process development overview: recovery yields and product quality (total viruses versus infective viruses (TP/IP)).
An analytical method for analyzing binding and elution of the viruses over a surface mimicking the ion-exchange matrix was developed by iBET scientists. A sorption rate model capable of quantitatively describing binding and elution of the recombinant baculoviruses and major impurities from dilute to overloaded conditions within a broad range of salt concentrations was subsequently validated (Figure 2A–2B).
A comparison of the adsorption of the major components of the baculovirus bulk onto an anion-exchange surface was conducted with this methodology. SPR operated as a pseudo-chromatographic tool utilized 1,200-fold less material (Figure 2C) than an analytical membrane adsorber unit for the estimation of adsorption isotherms.
Determination of the zeta-potential (a measure of the electrostatic equilibria) further elucidated the contribution of the different virus components to the interaction phenomenon during the ion-exchange process. The main species, product (infective virus particle) and product-derived impurities (dsDNA-, glycoprotein-, and envelope-deprived baculovirus particles), were isolated and correspondent zeta-potentials were measured through DLS.
The overall particle electrostatic interaction energy profile based on a fine-tuned electrokinetic model was able to distinguish the electrostatic properties of the infective virus particle from the major virus-related impurities. Scenarios such as Figure 2D allowed for prediction of the best operating conditions for promoting process selectivity.
Analytical tools based on SPR and DLS combined with in silico models grounded on fundamental knowledge can serve as relevant process predictors/scale-down models for addressing the useful design space more efficiently. The sensible selection of advanced downstream processes allied with rational design streamlines process development of such viruses and potentially other challenging biopharmaceuticals.
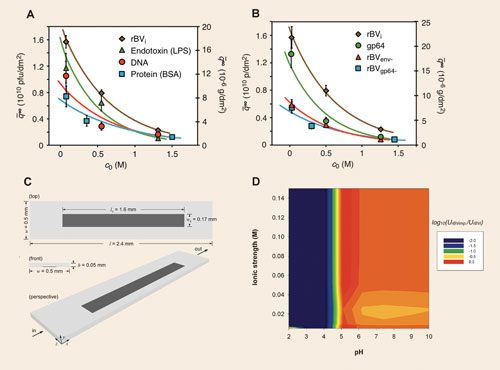
Figure 2. Comparison of adsorption capacity on DEAE sensor surface of recombinant baculoviruses (rBVi) and: (A) major process-derived impurities; (B) major product-derived impurities (baculoviruses depleted of gp64 protein, rBVgp64-, envelope, rBVenv-, and gp64 protein isolated). (C) Sensor surface geometry of a Biacore sensor chip. (D) Selectivity estimation between rBVi and major product-derived impurities. [A and B reproduced from Journal of Biotechnology, 148:171-181. C from Journal of Chromatography A, 1217:2032-2041 and D from Journal of Chromatography A, 1217:3754-3764 with permission of Elsevier]
Tiago Vicente, Ph.D. ([email protected]), was a DSP engineer in the animal cell technology unit (ACTU) of iBET/ITQB-UNL; he is now a DSP scientist at Redbiotec. Cristina Peixoto, Ph.D., is the head of DSP at ACTU. José P. B. Mota, Ph.D., is associate professor at FCT/UNL. Manuel J. T. Carrondo, Ph.D. ([email protected]), is the CEO of iBET and professor at FCT/UNL. Paula M. Alves, Ph.D., is the director of ACTU.


