Cancer immunotherapy is increasingly being evaluated as an approach to cancer treatment by harnessing the immune system to attack cancer cells. Both the adaptive and innate arms of the immune system play a pivotal role in a host’s defense against tumors.
CD8+ cytotoxic T lymphocytes (CTLs), which constitute a major component of the adaptive immune response, directly eliminate tumor cells by releasing cytolytic proteins such as granzymes, perforin, and granulysin, in addition to producing multiple cytokines. The ability to correlate T-cell biomarker expression/secretion with target cell killing is critical for tumor immunology studies.
Moving this research from the bench to the clinic is critically important, but reliable tools are needed to design in vitro assays that closely mimic activity in vivo. In this study, we use an impedance-based technology in combination with a bead-based multiplex flow cytometry assay to evaluate both the target and effector cells in a T-cell-mediated B-cell-killing potency assay (Figure 1). Real-time cell analysis (RTCA) technology with ACEA Biosciences’ xCELLigence® system provides us with a continuous readout of target cell viability, while flow cytometry enables the study of T-cell activation and functions by measuring cytokine and cytolytic protein secretion.
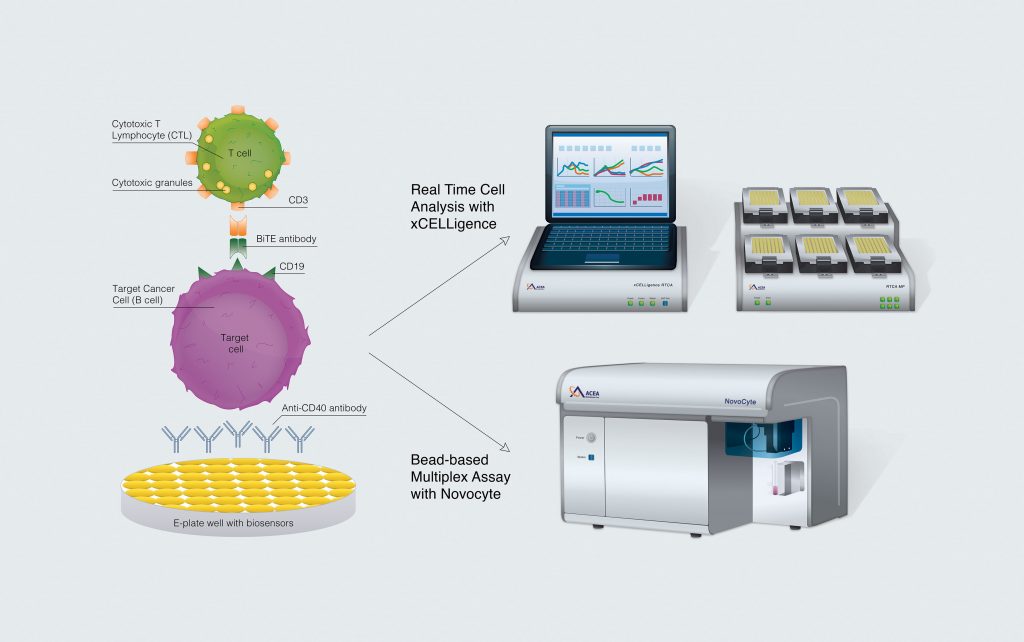
Representing a promising new class of therapeutics, bispecific T-cell engagers (BiTEs) harness the power of the adaptive immune response by enhancing the ability of CTLs to specifically recognize and eliminate tumors. CD19-BiTEs are designed to bind CD3 on CTLs as well as CD19 on cells of B-cell lineage, simultaneously activating T cells and bringing them in close proximity to the B cells, thus enhancing CTL effector function against various B-cell-derived tumors.
Here, the capability of CD19-BiTEs to enhance the cytotoxic effects of T lymphocytes on a B-cell lymphoma cell line (Daudi cells), was evaluated using two distinct assays. Target cell death was monitored using an real-time cell analysis impedance assay while secretion of cytokines and cytolytic proteins was qualified in a bead-based multiplex flow cytometry assay to evaluate the potency of the over multifunctional T-cell response.
Enhancing T-cell-mediated target cell killing
To demonstrate T-cell-mediated target-cell cytotoxicity on the xCELLigence platform, human T-cell killing of a B-cell lymphoma cell line (Daudi cells) was assessed. B-cell cancers are prominent immunotherapy targets because they are readily accessible within the bloodstream and are not affected by the microenvironment complexities or mixed tissue types associated with solid tumors. To help accelerate research in this area, ACEA Biosciences developed an xCELLigence Immunotherapy Kit for studying the cell killing of liquid cancers.
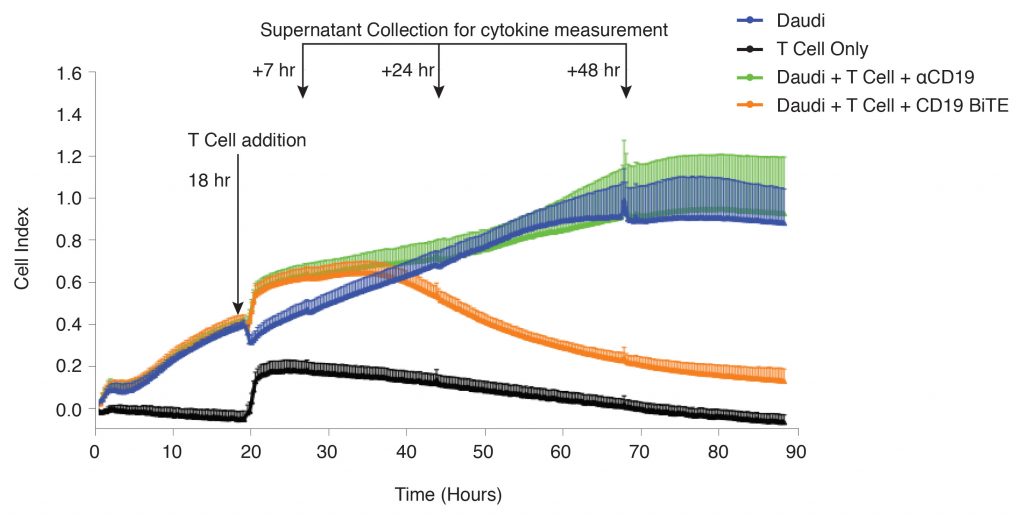
In this study, Daudi cancer B cells were immobilized on an xCELLigence E-Plate® precoated with an anti-CD40 tethering antibody. After the Daudi cells were seeded, T cells enriched from primary peripheral blood mononuclear cells (PBMCs) were added at an effector T cell to target Daudi cell ratio of 10:1. To measure if a BiTE can enhance T-cell killing, CD19-BiTE or an anti-CD19 antibody control was also added.
The xCELLigence technology utilizes proprietary E-plates which contain gold biosensors that are able to monitor cell behavior through impedance. The impedance signal of the Daudi cell monolayer was recorded every 15 minutes, and was reported using a unitless parameter called Cell Index. Uninterrupted growth and attachment of Daudi cells can be seen in wells with only Daudi cells (Figure 2, blue line), while no sustained impedance signal is generated from T cells alone, which can be subtracted as background (Figure 2, black line). Daudi cell growth is undisturbed by the addition of T cells and anti-CD19 antibody as a control (Figure 2, green line). However, a rapid decrease in Cell Index is observed with the addition of the CD19 BiTE in the presence of T cells, indicating that the Daudi target cells are being killed (Figure 2, orange line). This data demonstrates the capability of the CD19 BiTE to enhance T-cell-mediated cytotoxicity of B cancer cells.
Boosting cytokine and cytolytic protein secretion
To further study the effect of CD19-BiTE on T-cell activation and function, cytokine and cytolytic protein secretion was measured. Cells were cultured as described in Figure 2, and supernatant was taken at 7, 24, and 48 hours after the addition of T cells to measure 13 human proteins known to affect T-cell function with a bead-based multiplex assay on the NovoCyte flow cytometer (Figure 3). Consistent with our findings from RTCA, an increased secretion of CTL-associated proteins was observed. This data demonstrates that the presence of CD19-BiTE significantly enhances the production of cytokines and effector molecules that mediate and sustain target cell destruction. Seven hours after addition of effector T cells, cytokines specifically associated with a CTL response such as IFNγ, TNFα, and IL-2 increased 300-, 9-, and 10-fold, respectively.
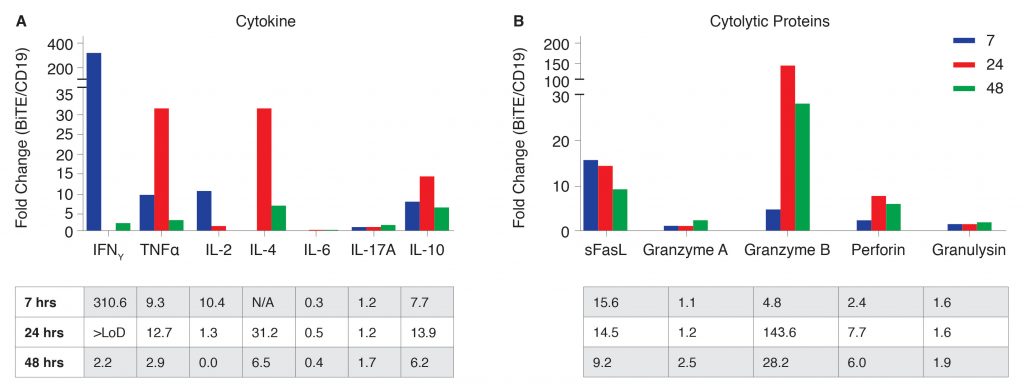
Secretion of cytolytic proteins such as sFasL, granzyme B, and perforin also dramatically increased by 24 hours after the addition of effector T cells, consistent with the CTL-killing response observed. This data demonstrates that CD19-BiTE enhances T-cell-mediated B-cell killing by increasing the production of cytokines and cytolytic proteins essential for a robust CTL response.
Summary
Here, we have coupled quantitative cell-killing assays with biomarker quantitation to provide an in-depth view of how CD19-BiTE affects T-cell-mediated killing of B cancer cells in a single workflow. The continuous monitoring of cell number, size, and
attachment quality using RTCA enables quantitative and kinetic assessment of the killing process. Linking this cytotoxicity data with quantitative analysis of cytokine and effector protein production allows for simultaneous analysis of T-cell activation and function. This
workflow, which integrates both cellular and protein analyses, advances current methods for research of cancer immunotherapy.
Lauren Jachimowicz, PhD, is an application development scientist, Aimee Chiavario is the senior marketing manager of immuno-oncology, Peifang Ye is a group leader, Garret Guenther, PhD, is the global support manager, Kenneth Chan, PhD, is the product manager, and Jeff Shurong Xue, PhD ([email protected]), is the director of marketing, at ACEA Biosciences, now a part of Agilent.