February 15, 2014 (Vol. 34, No. 4)
Steffen Panzner, Ph.D. CEO Lipocalyx
High-Tech Polymers Leveraging Viral Biophysics Can Enhance the Transfection of Nucleic Acids
Nucleic acid transfer is one of the central tools of the genomic era as it allows the manipulation of the genomic content of living cells in a predictable manner. In the context of in vitro applications, the virus-based transfer of genes (also known as transduction) is still considered the gold standard when it comes to the transfectability of different cell types.
Making viruses, however, is a laborious and complex process that requires significant expertise. In addition, viruses load larger pieces of nucleic acid and cannot be directly used to deliver oligonucleotides such as antisense, siRNA or miRNA constructs. As such, the advent of oligos, in particular siRNA, has created a huge demand for novel transfectants and greatly stimulated the development of synthetic vectors.
Viral and Synthetic Vectors Have Divergent Approaches
Independent from origin or cargo, any shuttle for a nucleic acid must at least offer three functional elements, namely (i) binding of the cargo, (ii) initial cellular uptake, mostly through the endocytic pathway, and (iii) escape from the endosomal compartment and release into the cytosol to enable activity. Other functions such as reverse transcription or genome integration are optional and do not necessarily define the core delivery event.
Viruses naturally fulfill these requirements, but do so through complex protein structures. The fundamental building blocks of synthetic vectors, on the other hand, are rather simple polycations. The charge complementarity offered by these not only facilitates binding of the nucleic acid cargo, but also promotes cellular binding. For the disassembly of the materials inside the cytosol, the strength of the interaction can be tuned by making adjustments to the charge density of the carrier.
This main technology feature of the synthetic carriers, however, is surprisingly different from its natural role model. Viruses do use cationic structures for the condensation of the nucleic acid, but do not expose a lot of these charged elements on their surface. The reason for this is due to the fact that cells, serum proteins, and mucus are all polyanionic structures; their negative charge represents the ground state in biology.
As such, the introduction of polycationic structures into such systems leads to undirected interactions and the unwanted formation of aggregates. This limited the use of the first synthetic transfectants such as polyethylenimine (PEI, a polymer), or DOTAP (a cationic lipid), to in vitro applications, where the cells to be transfected had to be adherent. In such a 2D arrangement, the transfection complexes could bind to the cell surface without triggering aggregation.
Conversely, these early transfectants were not compatible with suspension cells, could not penetrate tissues and had to be used under serum-free conditions.
As well as aggregation, the use of cationic structures suffers from another unfortunate problem. When exposed to low pH conditions such as those found in the cellular endosome, many of the polycations built into the carrier molecule exhibit an increase in charge density as their pKa value falls. This means that the transfection complex becomes even more hydrophilic inside the endosomal compartment, hindering membrane transition and having the exact opposite effect to that required to enable intracellular delivery.
Convergent Solution for Improved Transfection
The use of a new technology effectively circumvents the weaknesses exhibited by the portfolio of cationic carriers current available. Named Viromers, these polymer carriers still contain a polycationic core made from PEI, but these are densely substituted with hydrophobic and anionic side chains. The degree of substitution is so high that the resulting surface charge is almost neutral, such that the structures are fully compatible with suspension cells or serum.
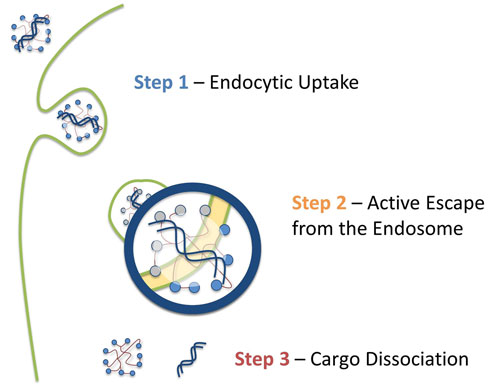
Viromers are taken up by endocytosis through an as yet unknown receptor and become gradually more hydrophobic during endocytosis as the pH is lowered, until at some point the transfection complex escapes from the endosome and enters the cytosol (Figure 1). The higher pH in this compartment prevents their transport back into the endosome.
In terms of mechanism, Viromers emulate an important part of the function of influenza hemagglutinin. This surface protein of the influenza virus encompasses a fusion peptide which is rich in hydrophobic and anionic side chains. If exposed to low pH during endocytosis, this fusion peptide will insert into the endosomal membrane of the cell. The same shift in solubility is exploited by Viromers, which carry nucleic acids along with them on their intracellular journey.
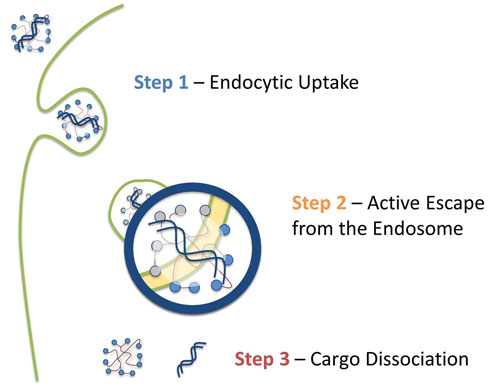
Figure 1. A schematic depicting Viromer uptake, followed by endosome escape and finally nucleic acid release.
More Effective RNAi and Improved Efficiency of Transformation
Viromer technology has shown promising results when used with a wide range of cell types, including many primary and suspension cells. A particularly strong improvement was observed when applied to phagocytic cells such as macrophages and monocytes, which rapidly endocytose objects from their environment as part of their normal function. While initial uptake in these cells is generally achieved using most transfection systems currently available, Viromers are particular effective at escaping the endocytic-lysosomal pathway of these cells to exert the desired influence on gene expression.
In one study recently carried out by Dr. Marc Bickle at the screening facility of the Max Planck Institute of Molecular Cell Biology and Genetics in Dresden, Germany, human macrophages were freshly isolated and transfected in a 384-well plate format. These were then treated using Viromers carrying siRNAs targeting the EEA1 gene, the knockdown of which is known to lead to a reduction in the number of cellular endosomes. When compared to a control siRNA, the number of endosomes observed was more than halved, even when using low concentrations of EEA1 siRNA (Figure 2).
The new technology also lends itself to plasmid transfection. In an analysis carried out at the Lipocalyx labs in Halle, Germany, three widely used cell lines in research and bioprocessing were transformed using Viromer Red, Viromer Yellow, and three other industry standard solutions, all carrying the pCMV-luc plasmid (100 ng total). This plasmid includes the luciferase gene, allowing transformation efficiency to be assessed by photometric assay. The expression levels of luciferase were 2–10 fold higher in the cell types transfected using Viromers, suggesting that gene expression and/or transformation was significantly improved using the new system (Figure 3).
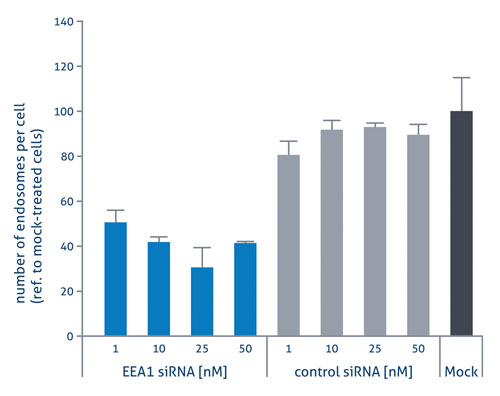
Figure 2. Transfection of primary human macrophages with siRNA using Viromers.
Conclusion
The Viromer technology represents a major improvement in the development of transfection agents. Based on a polymer that employs novel surface chemistry, it directly addresses the often neglected importance of enabling escape from the endosomal compartment. This is particularly relevant given that this step is ultimately responsible for the efficiency of nucleic acid transfer and the effectiveness of downstream applications such as RNAi and DNA transformation.
Viromers have already been used with a wide variety of common and challenging cell types. In addition, future developments are expected to extend the array of cells amenable to applications based upon nucleic acid transfection, opening up both new avenues of exploration and opportunities for optimization throughout the fields of research, drug discovery, and bioprocessing.
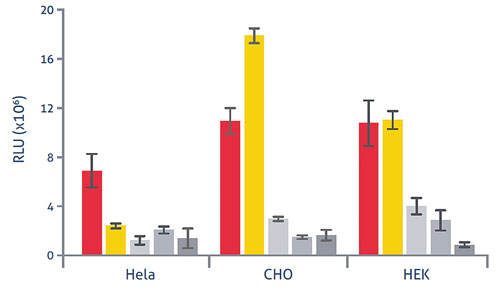
Figure 3. Improved transformation using Viromer Red and Viromer Yellow, versus other commercially available solutions (in grey).
Steffen Panzner, Ph.D. ([email protected]), is CEO of Lipocalyx.