July 1, 2010 (Vol. 30, No. 13)
Kathy Liszewski
A Handful of Companies Report Progress in Developing Next-Generation Antibacterial Agents
Multidrug-resistant bacteria pose a significant threat to global health. In 2004, the World Health Organization provided a list of the top 10 diseases for which there will be inadequate therapies in the near future. Infection by resistant bacteria topped the list, ahead of cancer and diabetes. In the U.S., methicillin-resistant Staphylococcus aureus alone annually infects more than 94,000 people and kills nearly 19,000—more deaths than from homicides, HIV/AIDS, Parkinson disease, or emphysema.
Additionally, resistant bacteria create an immense economic burden. One study suggested that such infections cost a staggering $21–34 billion in the U.S. alone. Although the need is rapidly increasing, there are growing concerns that the pipeline is withering away. A panel discussion at the “BIO International Convention” tackled these challenging questions, reviewed the current crisis, and discussed new solutions to create novel strategies for next-generation antibiotic agents.
“A perfect storm is closing in on antibiotic resistance,” suggested Michael Kurilla, M.D., Ph.D., director of the office of biodefense research affairs of the NIAID. “Although few companies are pursuing it, there is a large and growing unmet need for new antibiotics in the pipeline for drug discovery. There are many challenges that the industry faces, including regulatory uncertainties and market pressures. Another vexing issue is the ability of microorganisms to become resistant even to newly developed drugs.”
There are many types of emerging resistant bacterial strains. Some of the most common ones are gram-negative organisms such as Acinobacter, Pseudomonas, and others with extended spectrum beta-lactamases. Oral agents are needed for gram-positive strains as well. “Deciding which clinical indications to focus on is largely a matter of technical feasibility and market attractiveness.”
“While over ten years ago, there were numerous companies pursuing antibiotic drug discovery, today that number has shrunk to a handful. Help needs to come from several fronts. The FDA can assist by providing improved guidelines to help prioritize interests. Second, science must focus on and make strides especially in understanding serious gram-negative infections. Additionally, the market itself must become reasonable for development. Perhaps biotechnology can step up to provide new solutions and new approaches.”
Synthesis Strategy
Joyce A. Sutcliffe, Ph.D., senior vp biology at Tetraphase Pharmaceuticals, described the company’s approach for generating a new class of highly potent and safe antibiotics via a novel synthesis strategy.
“Making only slight synthetic changes in current drug scaffolds can result in big differences in terms of pharmacological properties. Our company is focusing on tetracyclines, which are historically, among the most successful classes of broad-spectrum antibiotics. They are also one of the most difficult to work with—only one new compound in this class has been approved in more than 30 years, largely because of chemical-modification constraints.”
“Our process was developed by Andrew Myers, Ph.D., chair of the department of chemistry and chemical biology at Harvard University. After 10 years of research, he developed a novel and completely synthetic approach to generate new tetracycline drugs and derivatives. In the past, chemically modified tetracycline analogs have been produced using solely semisynthetic or chemical transformation of isolated natural products. This severely limited the range of new drug candidates.
“Our core technology can modify tetracycline at any position. Our approach has provided us with a platform from which multiple complementary products have been generated, allowing us to identify potent compounds with the best spectrum and pharmacokinetic properties to meet medical needs.”
Tetraphase currently has one candidate, TP-434, that has completed Phase I trials; it is anticipated that it will enter Phase II studies later this year. Tetraphase has two additional IV/oral candidates for gram-negative infections or community-acquired bacterial pneumonia that are scheduled to enter Phase I trials in 2011.
“Our lead candidate TP-434 is effective against gram-positive, gram-negative, and anaerobic pathogens, except Pseudomonas,” Dr. Sutcliffe reported. “Because of its broad spectrum, it is expected to be utilized for complicated intra-abdominal infections, hospital-acquired pneumonia, complicated urinary tract infections, and acute bacterial skin and skin structure infections.”
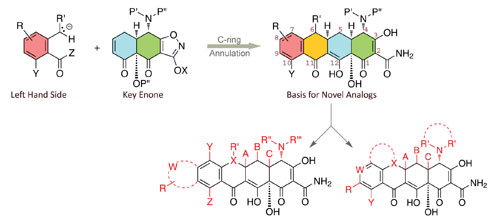
Tetraphase Pharmaceuticals prepares novel analogs with a convergent synthesis process that uses two advanced precursors, which allow modifications to be introduced at virtually any position on the resulting tetracyclic core. This diversity provides an enriched set of antibacterials with novel pharmacological properties.
Playing with Wild Cards
The treatment of hospital-acquired bacterial infections is costly to the healthcare system. George Miller, Ph.D., research fellow of Achaogen, noted that one recent study demonstrated that patients who developed a hospital acquired infection each incurred an additional $156,704 in costs, on average.
“Multidrug resistance strains are emerging as natural, evolutionary bacterial responses that ultimately defeat any antibiotic. Today, formerly effective antibiotics such as cephalosporins, carbapenems, aminoglycosides, and fluoroquinolones have been severely compromised by resistant strains. The wild cards are the rate of resistance and the virulence of pathogens.”
Achaogen is developing first-in-class agents, as well as enhancing previously successful approaches. The company’s lead product is a “neoglycoside” or novel aminoglycoside, ACHN-490, that entered into a Phase II trial in March. “A double modification to an existing aminoglycoside produced a neoglycoside.”
“ACHN-490 shows activity against multidrug resistant gram-negative pathogens and methicillin-resistant Staphylococcus aureus in preclinical studies. It is a broad-spectrum IV infusible therapy.”
Dr. Miller said that the compound “appeared safe and well tolerated in Phase I trials. Animal studies provided encouraging data that the compound was effective in murine lung tissue. It also demonstrated a more reduced potential for ototoxicity than comparator drugs in a guinea pig model.”
Overall, Dr. Miller sees ACHN-490 as a front-line antibacterial, particularly useful in the critical-care arena. Achaogen has four additional antibacterial drug programs in preclinical development.
Designer Antibiotics
“We don’t discover drugs, we design drugs,” noted John Finn, CSO at Trius Therapeutics. Dr. Finn discussed the company’s two major strategies for producing antibiotic compounds: structure-based drug design (SBDD) and focused antisense screening technology (FAST).
“Using high-throughput structural biology, we rapidly generate multiple ligand-protein structures. We employ SBDD tools to enhance drug properties, spectrum, potency, and target inhibition. The FAST platform consists of a set of bacterial strains that contain antisense DNA fragments that can be regulated to inhibit production of the target protein.
“Compounds that act on the protein downregulated by the particular FAST strain require significantly lower concentration of the test compound to inhibit growth. These strategies are a powerful approach for natural product discovery.”
Dr. Finn provided an example of Trius’ methionyl-tRNA synthetase (MetRS) program. “The aminoacyl tRNA synthetases are an underexploited class of antibacterial targets. These enzymes are critical for protein synthesis. Inhibitors represent a new class of antibiotic agents with good gram-positive activity. The traditional means for discovering antibacterial agents to this target yielded highly serum-bound agents. SBDD and FAST yielded potent gram-positive antibacterial agents that were effective in vivo within six months using one chemist.”
Another approach is to outsmart bacteria. “Smart bugs are essentially scanning bacterial genomes for multiple resistance mechanisms. One strategy to outsmart emerging resistance is to inhibit two critical bacterial targets with a drug. This strategy works particularly well when inhibition of the targets is synergistic. Synergy is an underrated tool.”
Trius has discovered agents that inhibit both MetRS and bacterial dihydrofolate reductase, an enzyme critical for bacterial growth and replication. “Combining activities in a single compound offers solutions to several antibiotic issues,” Dr. Finn said. “In the future, we expect to see an increased emphasis on synergistic drugs that can be used in reduced amounts, and thus, provide a higher safety profile. They can also yield a wide spectrum of activity and more easily avoid resistance.”
Unlocking the Ribosome
Rib-X Pharmaceuticals uses structure-based drug design targeted at the ribosome to design novel antibiotics, according to Erin Duffy, Ph.D., vp of structure-based drug design. Because it can delineate how drugs fit into ribosome pockets, 3-D structural modeling facilitates a practical understanding of the binding properties of antibiotics. It also reveals how mutations confer drug resistance. The derived information allows more rational design of novel chemical scaffolds that overcome bacterial resistance.
Ribosomes translate genetic information into proteins. Soil microorganisms have evolved the ability to produce antibacterial compounds that can block ribosome function. Some of these products have been converted into useful antibiotics that are currently employed commercially. Thus, Mother Nature has validated the ribosome as a suitable target.
According to Dr. Duffy, structural modeling can provide a clear picture of how each atom of the antibiotic molecule contributes. “In the case of the ribosome, it revealed for the first time how these different antibiotics engage the ribosome. Modeling demonstrated that they do it in adjacent but not overlapping ways. As such, they carve a completely new, validated, and enormous binding site for rational design.
“Our structural modeling, combined with proprietary computational approaches, allowed us to qualify and quantify high-value interactions these antibiotics make with the ribosome. Selecting any one of these key interaction sites as a starting point, our approach allowed us to build de novo completely new antibiotic scaffolds that take advantage of these interactions.
“Listening to the biology, our structure-based design approach can then use the surrounding areas to build-in properties that allow access to bacterial membranes, that afford efficacy in animal models of infection, and that show good safety profiles.”
Rib-X has three core programs: an empiric first-line treatment, Delafloxacin (a new drug application expected in 2014); Radezolid, a next-generation oxazolidinone (NDA in 2015); and, Rx-04, a broad-spectrum antibiotic that represents a completely new chemical class, active against resistant gram-negative strains (IND 2011).
“Delafloxacin provides simplified management of complicated infection. Its broad spectrum of coverage and excellent safety profile allows delafloxacin to be utilized as an empiric therapy that does not further compromise ill patients.
“Additionally, a second product in Phase II trials, Radezolid provides enhanced coverage, safety, and dosing as compared to linezolid. It concentrates in immune cells and is preferentially delivered to the site of infection. It also shows efficacy at low once-daily doses.”
The battle of the bug will likely continue for the near future. Trying to outsmart the crafty, adaptive microbes will require more emphasis on drug development such as the creation of novel synthesis strategies, identifying helpful drug synergies, and finding underexploited targets.
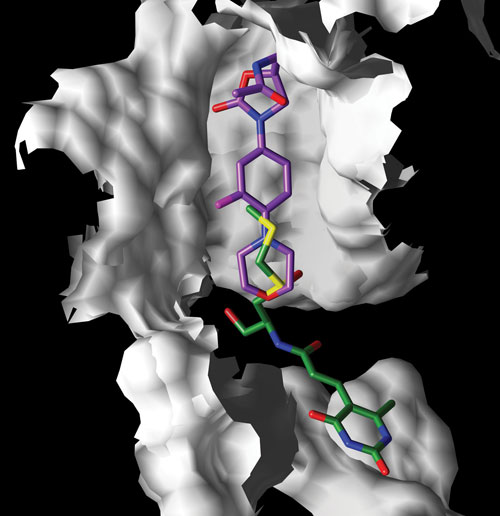
Rib-X Pharmaceuticals uses structure-based drug design targeted at the ribosome to design novel antibiotics. Linezolid (Zyvox, in purple) and sparsomycin (strong ribosomal binder, in green) are shown in their bound orientations in the ribosome. It was the revelation of the juxtaposition of their binding sites that suggested a validated way to build a next-generation oxazolidinone.
Sidebar: Notes from the Floor at BIO 2010
> Abbott continues to expand its biologics manufacturing capabilities for commercial manufacturing and clinical supply of drug substances, according to Ralph Lambalot, plant manager, biologics production, Abbott Bioresearch Center. Recent capacity expansions at its Barceloneta, Puerto Rico facility include an additional 12,000 L bioreactor for commercial supply of Humira, as well as the introduction of small-volume parenteral filling capabilities. The addition brings to the Puerto Rico facility the potential to readily expand to two trains with a combined 60,000 L capacity in the future.
In October, Abbott will bring its first fully disposable manufacturing suite online with the introduction of a 500 L cGMP manufacturing train using all single-use systems at its Worcester, MA facility. The disposable suite will enable Abbott to further accelerate clinical-development timelines with rapid changeovers for new product introductions, added Michelle Calhoun, commercial operations, marketing. The addition provides a fifth cGMP manufacturing train to the multiproduct facility’s existing four trains equipped with 3,000 L and 6,000 L bioreactors.
> Jim Hamby, Ph.D., vp of business development at Ash Stevens, reported that the company is in the early phases of a multiyear expansion that will add new capacities and capabilities to the company’s manufacturing plant in Riverview, MI. Upgrades will include new process-development laboratories, a high-potency drug development and manufacturing containment suite, a state-of-the-art materials-handling and storage facility, and a large-capacity reactor suite, among other enhancements.
“Our expansion is fueled in part by an emerging trend toward more innovator small molecule drug development and manufacturing staying onshore,” said Dr. Hamby.
> Lumigen, a Beckman Coulter company, recently introduced its HyPerBlu chemiluminescent substrate that detects hydrogen peroxide. According to the company, the substrate, which is a one-component solution with enhanced sensitivity and stability, is useful for research purposes and is amenable to high-throughput screening (HTS).
The company also has a new Sparcl™ technology that is a peroxidase-induced chemiluminescent immunoassay system to detect close proximity of antibodies and their analytes. Sparcl is designed for research purposes and it too is appropriate for HTS. According to Anthony Gaglio, product manager, strategic marketing, Lumigen is developing its own instrumental detection system for this flash chemistry-based assay, which is expected to be launched next January.
> Genetic Analysis works on molecular diagnostics and, in particular, creating “gut profiles” of bacterial flora. Applications for its technology include not only developing new drugs but also assessing the effect of drugs on the gut. Morten Isaksen, Ph.D., CEO, suggested that bacterial molecular diagnostics is a part of the growing trend for personalized medicine.
The company’s initial offering is GA-map™, an array technology that profiles a patient’s gut microbiota, providing screening of 100+ samples/day. The company currently is finalizing tests for infants and expects to move into adult screening tests later this year. Another application is to use these tests for research purposes to compare normal and diseases profiles. The company is looking for collaborators in these areas.
> iSentio also specializes in molecular diagnostics, but for mixed bacterial sequencing from clinical specimens. According to May Kristin Roen, CFO, the company’s new software tool, RipSeq®, has the ability to detect and identify bacteria directly from clinical samples by broad-range PCR targeting the 16S rRNA gene and DNA sequencing (direct 16S rDNA sequencing).
With this approach, an answer can be obtained in as little as 5–8 hours in contrast to traditional culturing approaches that take 2–7 days, she pointed out, adding that advanced algorithms eliminate the need for culture and manual separation of the bacteria prior to sequencing. Roen said this makes direct sequencing relevant for a broader range of clinical specimens including those from abscesses and pleural fluids.
The company is focusing on increasing the number of bacterial strains identifiable by sequence analysis. Several laboratories are currently using or testing the software application, and iSentio already has customers in seven countries including the U.S. and Germany, continued Roen. The company expects to open the first office in Asia in 2011.
> PCI Biotech has targeted the development of a novel photochemical drug-delivery technology for use in cancer therapy and various diseases. When illuminated, the introduced photochemical enhances the local effect of drugs because it releases biologically active drugs that are trapped in endosomes. The firm started clinical trials in August 2009 and has so far seen complete regression of all treated tumors, reported Per Walday, CEO. The company is focusing on cancer therapies for the head and neck.







