July 1, 2012 (Vol. 32, No. 13)
David Daniels, Ph.D.
Significant progress has been made in the effort to overcome the technical challenges inherent in measuring biological phenomena in single cells. This progress has been made possible by applying approaches from very different fields of study.
The goal in any biological study is to develop a method that allows you to “see” what is going on under physiological conditions without perturbing the system by the use of large labels or probes. With that in mind, Manlio Tassieri, Ph.D., Royal Academy of Engineering research fellow at the University of Glasgow, has developed a simple and noninvasive experimental procedure to measure the linear viscoelastic properties of cells, which he described at Select BioSciences’ “Single Cell Analysis” conference.
How can rheology, the study of the flow of fluids, be used to develop models describing complex systems like cells? By applying rheology principles to the study of deformations occurring at micron length scales and with µL volumes, Dr. Tassieri and his group have contributed to the field of microrheology by moving their research focus from in vitro models to in vivo systems like living cells. In order to monitor the physiology in cells and get a sense of the underlying cytoskeleton, the team attached an antibody-coated 5 micron bead (anti-CD4) to a Jurkat cell resting on a coverslip.
The team then tracks the movement of the bead in relation to the larger lymphocyte by video monitoring. In the test system, movement of the bead was influenced by the combined action of both the cell’s cytoskeletal activity and the thermal fluctuations of the fluid molecules within the media around the cell. For example, the response to a hypo-osmotic shock could be followed by tracking the bead.
“Osmotic regulation is fundamental to homeostasis of the cell as documented in the literature. Using our microrheology method, we are able to show that when the cell is moved from iso- to hypo-osmotic conditions, the cell swells and then stiffens up to 300% of its normal rigidity,” explained Dr. Tassieri. “This rigidity is based on bundles of actin filaments realigning, leading to the re-organization of the cytoskeleton. Then over a period of minutes, the cell relaxes.”
Similarly, the response to the addition of blebbistatin, a drug that blocks muscle contraction via interference with the actin/myosin interaction, can be monitored in the same way as shown for osmotic shock. The approach taken by the Tassieri lab is robust and reproducible, when compared to assays using techniques such as magnetic tweezers, optical tweezers, and atomic force microscopy. Further, the method has the advantage of revealing changes over a wide range of frequencies to a high level of accuracy.
Based on its simplicity and label-free nature, this application of microrheology could prove to be a valuable addition to studies that address cellular physiology under different pathological states.
Spectroscopic Analysis
Scientists at the University of Manchester, led by Peter Gardner, Ph.D., reader in the School of Chemistry and Analytical Sciences, are utilizing infrared (IR) spectroscopy for the analysis of cancer stem cell subpopulations. The application of IR spectroscopy is a well-established methodology in analytical labs for detection of impurities in chemical substances.
“IR spectroscopy as applied to the complexity of single cells is a problem that must be approached with a different mindset. You need to look at the spectral patterns not as a series of peaks assigned to different molecules, but rather look at the whole pattern like a fingerprint,” explained Dr. Gardner.
“A major breakthrough for us came a couple of years ago when we overcame the problem of excessive spectral distortion due to scattering, in particular due to a phenomenon known as resonant Mie scattering (RMieS). Scattering had more to do with the morphology of the cells than the biochemistry. That is, rounded cells showed high scattering, flattened cells showed less. We developed scattering correction algorithms, which meant that infrared spectroscopy can now robustly be used to study single cells.”
One project in the Gardner lab is the study of cancer stem cells, a subpopulation of cells in solid tumor tissue that are more resistant to chemotherapy and believed to be responsible for cancer recurrence. As a model system, the lab uses a renal cell carcinoma cell line (2245R) where the stem-like cells of interest make up less than 5% of the entire population. In the absence of specific cell markers, the team has exploited the observation that these stem-like cells overexpress the multidrug-resistance (MDR) pump.
Using a Hoechst dye efflux assay, the subpopulation of stem cells can be FACS sorted based on their lack of cytoplasmic dye and cytospun onto slides for spectral analysis using an imaging infrared spectrometer. IR spectroscopy signatures can be captured from thousands of cells in a rapid and reproducible data output. The spectral signatures can reliably differentiate cell populations—for example, differentiate between cells that overexpress the MDR pump from those that don’t.
The spectral contrast comes from the cell makeup and is not dependent on stains or dyes. Routinely, it is possible to overlay an optical image of the cells with their IR spectral image so that each cell and the associated spectrum can be spatially located. The IR spectral signature, corrected for the scattering artifacts, registers only the absorption events that provide information about the cellular chemistry including glycogen levels as well as lipid and phosphate profiles.
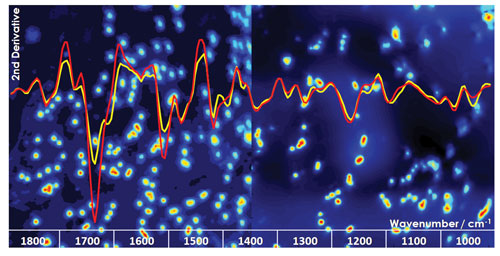
University of Manchester researchers are utilizing IR spectroscopy for the analysis of cancer stem cell subpopulations. Infrared hyperspectral image of cancer stem cells (left) and a second derivative infrared spectrum (red) and normal cancer cells (right) with the second derivative infrared spectrum (yellow).
Signaling
Signal transduction based on a ligand binding to a receptor on the cell membrane and the subsequent cascade of phosphorylation that ultimately leads to gene expression in the nucleus that happens over a period of minutes is well understood. But what about the early events, the subsecond molecular processes that occur when the ligand binds the receptor?
To capture these events, Jonathan West, Ph.D., and Ya-Yu Chiang, Ph.D., of the miniaturization group at ISAS, Dortmund, Germany, have developed a microfluidic system for the rapid treatment of single cells. The key is to use a two-step deterministic lateral displacement (DLD) technique that “bumps” cells from one laminar stream into a stream for receptor stimulation with ligand molecules and subsequently bump them into another stream for reaction quenching.
The incubation time is precisely defined by the cell speed and the distance between the DLD switching elements. This design avoids the need to mix the ligand with the cells as that process takes too long (seconds) to effectively capture the initial events.
The West team is able to record the signal transduction dynamics with unprecedented temporal resolution involving millisecond switch times each with microsecond precision.
“We use the insulin-like growth factor receptor (IGFR) as a model for this analysis because the receptor is already dimerized in its active state on the cell membrane. This ensures the molecular states we capture are synchronized and not hidden within the variable diffusion timescales required for dimerization upon ligand stimulus,” commented Dr. West.
“Dr. Chiang has perfected the microfluidic timing and built an envirostat system that controls the normal cell culture conditions (37°C and 5% CO2) to maintain the integrity of the cells from syringe to microfluidic stimulation and reaction arrest. We can interrogate the status of the kinase center and determine the nature of the switching on process by comparing data from lengthy ligand treatments and from various IGFR mutants. Mechanistically, does the center work like a light switch or is it more like a domino effect from one phosphorylation site to another?”
Now that the team has optimized the system and demonstrated proof of concept, some of the interesting biological questions are just now being addressed. Is there cell-to-cell variation in stimulus patterns? Are there reaction intermediates that haven’t been seen before?
Beyond the study of early signaling, the system is amenable to the investigation of a huge variety of fast cell-surface interactions. Dr. West hopes the microfluidic technology will prompt biochemists and cell biologists to ask new questions.
qPCR
Can you extrapolate the analytical results from the study of a cell population to what is going on in a single cell? As technology and analytical methods improve, scientists are starting to ask those questions and coming up with some surprising results.
At Fluidigm, Kenneth Livak, Ph.D., senior scientific fellow, has focused on meeting the challenges of analyzing single-cell gene-expression data. Practical steps for analyzing single-cell qPCR data had to be developed because the stochastic nature of eukaryotic transcription at the single-cell level meant that conventional methods for analyzing qPCR data often do not apply.
The company has developed protocols and developed an instrument, the BioMark™ HD, that enables qPCR analysis of transcripts from single cells. In population studies the data yields an average value rather than a real measure of individual cells.
The data can be misleading because this average is dominated by a small number of cells that have a high number of transcripts. Consequently, average measures of gene expression don’t give the real picture of what’s going on in the single cell as most cells in a population, even a homogeneous population, have a substantially lower number of transcripts that the average value.
“Single-cell gene expression is intrinsically noisy,” noted Dr. Livak. “This is based on the observation that eukaryotic transcription occurs in pulses with bursts of transcription interspersed with inactivity when transcripts decay. Consequently, there is significant variation from cell-to-cell that can range as much as 10- to 1,000-fold for every gene we look at.
“Normalization to housekeeping genes doesn’t help, as these housekeeping genes can vary as much as the genes we’re studying. We find that it is best to use ‘unnormalized’ data that actually is normalized on a per cell basis.”
More specifically, Dr. Livak reported that the solution is to look at enough genes in enough cells to apply a multivariant analysis to get a robust signature as to what is going on at the level of single-cell gene expression. A multivariant analysis, either hierarchal clustering or principal component analysis, allows you to look at the pattern of all genes in the study to get an understanding of what is happening at the individual cell level.
At Fluidigm, the approach is to take FACs-sorted tissue culture cells and plate them individually directly into lysis buffer in wells in a standard 96-well PCR plate. Reagents are added and the plate is put in a thermal cycler for the multiplexed reverse-transcriptase step. Ideally, in this multiplex preamplification step, sufficient cycles are run so that single cDNA molecules will generate at least five molecules per qPCR reaction chamber in the next step.
The cDNA pools are transferred by eight-channel pipetter to one side of the microfluidic chip. On the other side, singleplex PCR primers for the 96 genes to be analyzed are added. The chip is then put within the Biomark HD instrument where all pairwise combinations are mixed. The PCR reactions are run and analyzed in real time within the instrument, yielding 9,216 data points. The data output is presented as a histogram that shows the number of cells with each expression level bin for all the genes in the study.

According to Fluidigm, the Access Array IFC is the first commercially available microfluidic device that allows the user to recover the PCR product. It automatically assembles 2,304 unique PCR reactions, each reaction a combination of one of the 48 samples and one of the 48 primer inlets. Post PCR the product of 48 reactions per sample is harvested for downstream sequencing.
Genomics Research Center
Convinced that this approach to gene expression in single cells will give new insight to biological phenomena, Fluidigm, in collaboration with the Broad Institute, recently announced the formation of a new single-cell genomics research center to be housed at the Broad Institute, where Dr. Livak will serve as the alliance manager. In this role he will oversee research projects at the center to make sure that while they deliver scientifically, they also help to develop protocols and identify opportunities that can be used and shared with the wider scientific community.
Fluidigm said the Broad Institute is the first of what could be many institutes around the world that will join the firm in its quest to push the envelope on what is known about biological phenomena at the single-cell level. The center will focus on the acceleration of the development of research methods and discoveries in mammalian single-cell genomics, as well as serve as a hub for collaboration among single-cell genomics researchers in pioneering fields including stem cells and cancer biology.
In the end, these learnings may help us, by analogy, to understand the forest for the trees.



