October 15, 2010 (Vol. 30, No. 18)
Hexapeptide Kits Useful for Many Sample Types
One of the challenges in proteome analysis is that samples are often dominated by a relatively small number of high-abundance proteins whose presence can obscure less-abundant proteins and limit the capacity and resolution of the separation technique(s) employed. This is especially the case for serum and plasma where some 20 proteins constitute more than 98% of the protein mass, according to Aran Paulus, Ph.D., R&D manager of new technologies and proteomics applications at Bio-Rad Laboratories.
Many other tissues and cells also have high percentages of specific proteins, Dr. Paulus adds.
In a poster entitled “Enriching Low-Abundance Proteins Using ProteoMiner™ Protein Enrichment Technology,” S. Freeby et al. at Bio-Rad discuss the removal of highly abundant proteins by two different approaches. The first is immunodepletion, which is used to remove species-specific proteins in serum or plasma.
“Alternatively, one can use a library of combinatorial hexapeptides to bind all possible proteins in a complex mixture,” notes the Bio-Rad authors. “In this approach, which is independent of the sample source, the concentration of high-abundance proteins is reduced, and medium- and low-abundance proteins are enriched.”
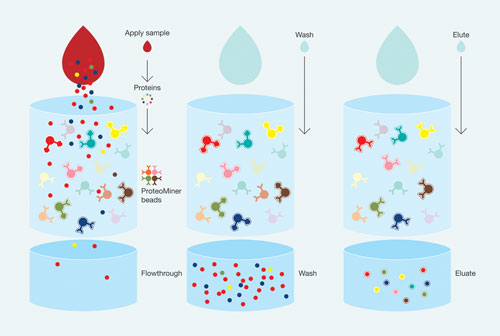
ProteoMiner technology removes a large proportion of the high-abundance proteins in a complex mixture, facilitating detection of low-abundance protein biomarkers, according to Bio-Rad. Users mix their sample with a diverse library of hexapeptides bound to chromatographic beads. High-abundance proteins will rapidly saturate their hexapeptide ligands (red and yellow beads), and excess protein can then be washed away, while low-abundance proteins will remain bound to their ligands (pink and teal beads) and concentrated on the chromatographic support.
Kit Format
Bio-Rad markets the hexapeptide technology in the form of its ProteoMiner enrichment kits. Since this technology does not rely on antibodies, it can theoretically be applied to multiple sample types.
The Bio-Rad research described in the poster was designed to examine the applicability of ProteoMiner protein enrichment kits for the reduction of high-abundance proteins and enrichment of medium- and low-abundant proteins from heart muscle, HeLa cells, and E. coli protein samples. The scientists discussed the conditions for protein extraction, treatment with ProteoMiner beads, and analysis with 2-D gel electrophoresis.
The group concluded that the technology, first developed for the reduction of high-abundance proteins in serum and plasma, does work with tissues, cell lines, bacterial, and plant samples. They said they found a significant reduction of the top high-abundance proteins in each sample set, as confirmed by mass spectroscopy identification.
In addition, “heart and HeLa cell proteins that have been extracted under denaturing conditions can be used successfully with ProteoMiner protein enrichment kits,” they wrote, adding that “ProteoMiner bead treatment enriches a unique population of proteins not apparent in the untreated samples.”
Because of the removal of greater than 95% of sample protein mass, protein spot numbers for heart, HeLa cells, and E. coli increased 14%–28% following ProteoMiner bead treatment, the team reported.



