January 1, 2008 (Vol. 28, No. 1)
Matthew Daley
Jibin Hao
The Integritest 4 Was Assessed Using Oversize Volumes to Determine Accuracy of Results
Sterilizing-grade filters are designed to reproducibly remove microorganisms from the process stream producing a sterile effluent. To ensure product sterility and regulatory compliance, membrane filters must be integrity tested before and after filtration of a drug product to ensure that filters with the correct pore size were used, the filter was installed properly, and the filter was free of defect.
Because filter integrity testing is a critical step in the manufacturing of sterile drug products, it is required by regulatory bodies. Users can conduct bubble-point, diffusion, enhanced bubble-point, and HydroCorr tests on disk, cartridge, and TFF filters as well as virus and asymmetric membrane filters.
One of the most widely used integrity tests is the bubble-point test. This is a sensitive visual technique, based on the fact that liquid is held in the pores of the filter by surface tension and capillary forces. The minimum pressure required to force liquid out of the pores is a measure of the pore diameter.
If the filter is manufactured out of spec, a stream of air bubbles will appear downstream of the filter membrane before the acceptable bubble-point pressure is reached. The bubble-point test can detect minor filter defects and out-of-size pores and correlates with the bacteria-passage test.
The diffusion test is based on the fact that gas will diffuse through the pores of a fully wetted filter. Pressure is typically applied at 80% of the bubble-point pressure of the filter. The overall rate of diffusion is proportional to the surface area of the membrane in the filter.
The flow rate is measured on the upstream side of the filter with an automated instrument. There would be a dramatic increase in gas flow at this pressure if these were damaged membranes, wrong pore size filters, ineffective seals, or system leaks.
As larger processes and batch sizes become the norm, it is necessary to have the technology to integrity test at large volumes. Until now, however, the instrumentation hasn’t been robust enough to handle large housing volumes.
One of the main challenges of integrity testing is accurately and reliably testing large filter systems and large upstream volumes. When working with such large volumes, rewetting the filter after each test requires a large amount of fluid, which is both costly and time consuming. Without rewetting the filter between tests, the accuracy of the test can be affected.
Millipore (www.millipore.com) assessed the Integritest® 4 (IT4) automated integrity-test instrument using large volumes to determine accuracy of results. The IT4 was challenged with a worst-case scenario, which involved testing a 12×30 filter housing with greater than 100 liters of upstream volume. Multiple tests were performed on asymmetric pore filters without rewetting between tests.
Materials and Methods
A 12×30-inch housing capable of fitting 12 0.2 µm hydrophilic filters was provided by Millipore. The upstream volume of the housing was appended, so that the upstream void volume was at least 110 L. The filters were wetted at ~150 LPM for 10 minutes with ~10 psi back pressure. The wetting was performed in a recirculation mode. The filters used in the study were 0.22 µm polyethersulfone membranes with asymmetric pore size. After the filters were wetted, five diffusion tests were performed with the Integritest 4.
The results from the tests were recorded and all test strips saved. The filters were not wetted in between tests. It is recognized that in practice, filters are always wetted prior to an integrity test. The purpose of this test, however, was to compare integrity tester measurements with offline measurements without rewetting. Offline measurements were performed using a calibrated mass flow meter with 1,000 mL/min flow rate range (Sierra Instruments).
Results
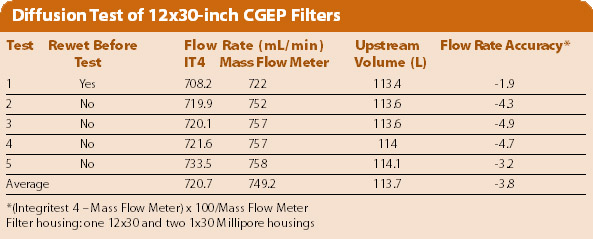
The Table shows a sample of the test results when the filters were not rewetted between the test. The accuracy specification for the Integritest 4 is that the flowmeter measurement and the Integritest measurement are within 5% of each other. As shown in the Table, that accuracy was met with upstream volumes as high as 114 liters. It is important to note that this accuracy was achieved without any ancillary equipment.
Table
Conclusion
Today large batch sizes of pharmaceutical drug products are common. Increases in batch sizes, however, can lead to challenges in filtration, due to the viscosity as well as solids and particulate content of these batches. This trend leads to increased filtration costs as well as a high demand to maintain the same or better filter performance and reliability.
Therefore, it is important to also maintain the high level of accuracy of the integrity testing methods used. Based on this case study, integrity testing filters at large volumes can be done accurately.
Millipore’s Integritest 4 automated integrity test instrument is based on pressure decay technology. The accuracy of the Integritest 4 instrument’s flow rate is dependent on the accuracy of measuring upstream volume, which is affected by the fluctuation of gas temperature in the upstream volume.
In the past, the upstream volume of the test without rewetting was 5–10% lower than that with rewetting. The root cause of this difference is that some of the pressure decay data for the test without rewetting are affected by the temperature during sizing upstream volume. This is fixed by excluding this data in the upstream volume calculation. As shown in the Table, the upstream volume is almost constant, whether the filters are rewetted or not.
Matthew Daley is field marketing
specialist, and Jibin Hao is consulting engineer at Millipore. Web: www.millipore.com. Phone: (781) 533-2558. E-mail: [email protected].







