October 1, 2010 (Vol. 30, No. 17)
William Downey
Cindy Liu
Jennifer Hartigan, Ph.D.
Contract Manufacturers Bank on New Initiatives to Boost Capabilities
In August, U.S. Senator Michael Bennet (D-CO) introduced the Drug Safety and Accountability Act of 2010. This bill is designed to improve regulatory oversight of drug manufacturing facilities and improve related quality standards and monitoring.
With increased government scrutiny, it is not a coincidence that quality is the top-ranked criteria for CMO selection, according to findings from HighTech Business Decisions’ “Biopharmaceutical Contract Manufacturing Report: Expanding Markets, New Capacities and Improved Performance” for which directors from 48 pharmaceutical and biotechnology companies and 29 biopharmaceutical CMOs were interviewed.
Biopharmaceutical contract manufacturers continue to dedicate resources to improve quality and efficiency to meet the increasing demands from their clients and regulatory organizations.
As a result of strict regulatory oversight, once a standard operating procedure is validated and yields drugs acceptable to the FDA or EMEA, companies are reluctant to make any process changes or incorporate innovations. In response to this situation, the FDA issued a draft guidance document, PAT—a Framework for Innovation Pharmaceutical Manufacturing and Quality Assurance. FDA hopes that this guidance will help pharmaceutical manufacturers design, develop, and implement efficient tools for use during product manufacture and quality assurance.
Many companies have followed the PAT guidelines and designed innovative quality improvements into their processes. For example, Boehringer Ingelheim (BI) developed the BI-Purification Excellence (BI-PurEx) strategy to shorten development time while improving process understanding during early development phases.
The strategy is designed around preferred process steps and takes advantage of a set of highly sensitive analytical tools and automated screening technologies to efficiently develop each unit operation in a downstream process.
According to Barbara Esch, director of global marketing and business development coordination, BI-PurEx has resulted in a “more lean development process to reduce timelines in early development. In addition, through the integration of analytical assays and RAPPTor® high-throughput screening, process knowledge is gained throughout process development to maintain a critical focus on future market supply.”

Boehringer Ingelheim developed the BI-Purification Excellence strategy to shorten development time while improving process understanding during early development phases.
Also following PAT guidelines, RecipharmCobra Biologics recently implemented new analytical approaches for monoclonal antibody development. RecipharmCobra successfully developed high-throughput platform methods for residual host cell protein, DNA, antibody titer, and antigen binding for early potency determination based on the Gyros™ system.
“These platform analytical methods are qualified so that they can be used upon verification under product-specific conditions for GMP release and for the stability testing of drug substances and drug products for early clinical trials,” explained Christel Fenge, GM. “This has allowed RecipharmCobra to reduce effort and time lines for analytical development early on in projects. One of the benefits for the customer is obviously the reduced cost of development associated with bespoke analytical methods and lengthy manual procedures.”
RecipharmCobra also has experience with PAT applied to QbD (quality by design), fostered by its history in product development. This expertise is of particular interest to biotechs, which RecipharmCobra actively supports in the appropriate design of product characterization studies to meet the requirements of the relevant clinical phase. “We consider this to be a competitive advantage and differentiator to other CMOs as we are able to offer a one-stop solution, from the gene of interest to GMP drug product accompanied by a comprehensive documentation and testing package,” Fenge noted.

A technician at RecipharmCobra Biologics prepares for a GMP manufacturing run.
The Merck BioManufacturing Network, which is composed of two Merck business units—Diosynth Biotechnology and MSD Biologics—builds in QbD in biologics development and manufacture by way of high level of process understanding.
As Stephen Taylor, commercial director of MSD Biologics, explained, “during process development and laboratory process characterization (LPC) studies, the aim is to acquire a good understanding of each unit operation and define operating windows, to ensure acceptable and consistent product quality.
“If a high degree of process understanding is gained, and the process is consistently operated within the defined windows, then theoretically there should be less need to incorporate PAT.” According to Taylor, this approach brings operational and delivery benefits to clients by enabling a relatively high throughput of different products.
PAT is also being used in process development and selected manufacturing campaigns at Merck. “During a typical fermentation, critical quality parameters such as pH, temperature, CO2 evolution, O2 uptake rate, and respiratory quotient are monitored in real time using probes and in-line mass spec analysis,” Taylor commented. PAT has been implemented during downstream processing, though to a lesser extent “where real-time monitoring has been used to determine when process steps have progressed to an optimal level,” he added.
CMC Icos Biologics has integrated analytical characterization of the protein produced during the clonal selection phase of cell-line development with its CHEF-1® expression platform, focusing on selection of clones producing the highest quality product in addition to selecting for productivity and viability.
Jenifer L. Wheat, vp of business development, said that “ensuring choice of a clone expressing protein that matches the specifically desired analytical profile is critical for second-generation processes and biosimilar development. Monoclonality is also assured through imaging of colony outgrowth with Genetix’ CloneSelect, providing higher confidence levels in clonality than traditional limiting dilution cloning.”
Meridian Life Science is actively evaluating and implementing PAT initiatives. Design control, environmental monitoring, process yield improvements, CAPA streamlining, risk management, and process mapping are a few of the initiatives being implemented.
“These processes are helping Meridian increase our competitiveness and fully meet the needs of our customers in domestic and international markets,” commented Alan Rich, director of regulatory affairs and QA/QC. “They are allowing Meridian to shorten lead times for new product releases, reduce costs, minimize headcount, improve quality, and reduce scrap.”

The Merck Production System, which is based on Toyota’s production system, is being rolled out across the company’s biomanufacturing network.
Lean and Six Sigma Principles
In an effort to do more with less, CMOs are using several analytical approaches to create a more efficient operating environment. The industry is turning to lean design methods to identify manufacturing inefficiencies and improve supply chain management.
Manufacturing bottlenecks can be identified through operational simulations and multivariate analysis, using inline sensors to generate process data in real time. To ensure success, the lean manufacturing concept not only needs commitment from the top management, but it is essential that the concept be part of the culture and ingrained in everyone’s daily activity.
Boehringer Ingelheim has been using Six Sigma and lean manufacturing tools to improve business and manufacturing processes for its biopharmaceutical operations since 2003. In order to gain maximal results and a continuous improvement culture, BI has a top-down and a bottom-up systematic methodology.
BI’s business process excellence (BPE) policy has the commitment of top management as well as every employee. “We defined clear functions and responsibilities, made a process analysis, mapped the most value-added processes, and fixed them in a process landscape,” Esch added.
According to Esch, “efficiency gained from Six Sigma and lean manufacturing tools included minimizing redundant process steps and reduction of wait and throughput times by more than 15 percent.”
Meridian is another company where the lean manufacturing concept is ingrained in its culture. All Meridian management is expected to lead a process improvement project. Project progress is reviewed with the division president on a quarterly basis. Process improvement is also expected of each employee even if they have not gone through formal lean or Six Sigma training.
“By improving and streamlining our manufacturing processes we have seen dramatic improvements to the top line and bottom line, less complaints, improved customer audits, less variability, time savings, and less scrap,” reported Lourdes Weltzien, GM.
Merck sites recently implemented the Merck Production System (MPS), which is based on Toyota’s production system and incorporates key principles of Six Sigma across the manufacturing sites. “Process development is closely integrated with manufacturing activities so we are carefully planning how to deliver the benefits of MPS in process development so that it strengthens rather than compromises the strong innovative culture we have in place,” Taylor commented.
In 2009, RecipharmCobra focused specifically on reducing lead times by applying visual planning as a tool to improve throughput, which led to a significant reduction in lead times for deviation close-out. “Clearly, for a CMO, standardization of business processes is an area where benefits can be generated, and this continues to be important to us in an environment where customer projects are unique,” said Dr. Fenge.
Besides the hard quantifiable savings from lean manufacturing tools, there are intangible benefits that translate to high success rates in process transfers, improved flexibility in manufacturing scheduling, and increased manufacturing robustness as reported by Boehringer Ingelheim. Dr. Esch also touted “soft factor” benefits including team spirit and improved communication between departments that can lead to better identification and elimination of process risks.
Biologic Conjugates
An important new area in biologics manufacturing is the development of biologic conjugates, made through genetic engineering of fusion proteins or conjugation of an additional compound to a protein after purification. Although this process adds a layer of complexity to an already complex manufacturing process, biologic conjugates can provide targeted therapy with improved efficacy and safety profiles.
These protein conjugates can be divided roughly into two types—one type is engineered fusion molecules that reduce immunogenicity and target enzyme activity, and the other type is chemically modifying protein drugs that improve biological half-life and targeted cytotoxic effects.
The cost of goods increases significantly with this type of manufacturing because of challenging cell-line development, product purification, and the complex chemistry of conjugating a chemical compound with a fragile protein without diminishing its biological activity. The problematic manufacturing required to produce conjugated biologics provides an opportunity for CMOs familiar with these complex processes.
One of the major issues related to biologics conjugates is the uniqueness of these compounds. “The applicability of platform approaches is limited and each conjugate has specific physicochemical properties, development, and production challenges. The absence of platform approaches makes this kind of development more expensive and scientifically more challenging. Customers may not fully appreciate the scientific and technical challenges and the associated costs of overcoming them,” explained RecipharmCobra’s Fenge.
One exception is the successful application of platform approaches to genetically engineered fusion proteins. Boehringer Ingelheim’s Esch explained that, “for half-life extension some proteins are expressed already at the genetic level as fusion molecules. The advantage of those technologies is the establishment of a lean and cost-effective large-scale manufacturing process, whereby, the molecular size of the fusion partner determines what expression technology might be best.”
Dr. Fenge explained that “for Fc-fusion proteins, RecipharmCobra has built on platform approaches developed for monoclonal antibody purification, particularly with regard to the protein A or G capture step.”
Merck has had similar success expressing fusion proteins. “In our experience, the principal challenge with fusion proteins is in the selection of an appropriate expression system; depending upon the exact nature of the conjugate, we have found that yeast-based systems are generally reliable production platforms for this class of biologic,” said Taylor.
Despite the use of well-characterized expression platforms for genetically engineered fusion proteins, these types of conjugates can be difficult to work with.
“Typically, titers tend to be rather low compared to monoclonal antibodies,” said Fenge. “This might be due to their complex and often ‘foreign’ structure compared to naturally occurring proteins; leading to aggregation, truncation, improper secretion, or other problems in the cellular process, which result in an improper conjugate structure.”
According to Esch, “soluble expression of toxin conjugates in various expression systems accounts for toxication of the respective production organism due to the toxicity of the fusion partner.”
The challenge of manufacturing chemically modified biologics reaches beyond the lack of platform approaches. Esch noted that “the chemical modification after initial expression and purification adds more process complexity and additional product characterization efforts.” Taylor added that “as far as protein-PEG conjugates are concerned, the main challenge is, generally, to ensure that the PEGylation chemistry is compatible with the biologic partner.”
CMC Icos Biologics’ Wheat echoed a similar sentiment with respect to toxic conjugates indicating that, “as chemically conjugated entities are typically cytotoxic and/or radioactive, conjugates present unique operational challenges for biologics CMOs. Typically, separate facilities are required for the chemical conjugation as well as for the subsequent purification of the conjugated molecule.”
Overcoming the challenges of chemically modified conjugates tends to be resource intensive and “requires an intimate collaboration and true partnership between the CMO and the customer,” noted Fenge. “In some cases, it might be necessary to work with the host cell line or embark on a protein-engineering exercise to make a functional conjugate at a reasonable cost of goods.”
Experienced contract manufacturing personnel provide specialized skills needed for complex manufacturing of conjugated biologics. “As a contract manufacturer, one has to keep several different technologies ready to match the technology to the customers’ needs. Boehringer Ingelheim has broad experience with various types of biologics conjugates,” said Esch.
“For example, to circumvent the problem of host toxication, production in recombinant E. coli in insoluble form is potentially a simpler, more cost-effective method that makes the manufacture of such toxic biologics conjugates economically viable.”
Merck has experience in the manufacture of fusion proteins and proteins conjugated with chemical moieties such as PEG, according to Taylor. “We have gained significant experience in this and usually rely on DOE (design of experiments) to identify optimal conjugation conditions.”
RecipharmCobra Biologics is also experienced with a variety of different conjugates including “classical” Fc-fusion proteins, novel two-domain fusion proteins expressed in CHO cells, and (Fab)2-enzyme conjugates expressed in E. coli involving chemical conjugation. Fenge commented that “protein-analytical expertise and broad capabilities are especially important to success, and it is critical early on in development to understand the compound and to be able to monitor potential product degradation or improper processed forms.”
Conjugated biologics are improved drugs. For example, chemically linking toxins to antibodies provides targeted rather than systemic drug activity. For half-life extension, genetic engineering and pegylation, “technologies open opportunities in the field of patient-friendly application forms,” said Esch.
Business Outlook and Opportunities
In addition to expanding its quality capabilities, the CMO industry continues to invest in capacity expansions. The CMOs interviewed for this article were generally optimistic about their business prospects.
Overall, CMOs are expecting revenue growth in the range of 25% to 30% from 2010 to 2011. This growth expectation comes largely from a combination of capacity expansion, increased demand for novel vaccine and biologic manufacturing, and expanded service offerings.
In April, Cytovance Biologics was awarded a biopharmaceutical development and manufacturing contract by Selexys Pharmaceutical for its Anti-PSGL-1 therapeutic agent. Cytovance is undertaking facility and personnel expansion to meet current and anticipated demand from customers. It has added a 1,000 L bioreactor to augment its 100 L and 500 L mammalian production suite and is expanding the bioanalytical laboratory and process development space.
Over the next 18 months, Cytovance is planning to expand its microbial manufacturing and aseptic fill-and-finish capabilities as well as add a 5,000 L bioreactor. According to Bill Dull, vp of sales and business development, “this strategy to increase manufacturing capacity and services is a direct result of current customers partnering with us to move beyond clinical-stage production and meet their commercial-scale manufacturing needs.”
In August, CMC Icos Biologics reported the completion of a new state-of-the-art disposables manufacturing facility at its Seattle site. The addition comes as part of a staged major expansion plan, which will add significant production capacity to its existing facility to accommodate growing demand for cGMP manufacture of biopharmaceuticals.
“CMC’s expanded service offerings include analytical and formulation services as well as the development of unique solutions for clients through various combinations of our expression platform, cell-line development, proprietary medium, and analytical services, including evenly matched upstream and downstream scales in our new single-use disposable bioreactor facilities,” he added.
Meridian Life Science reports seeing a great deal of interest especially in novel vaccine preparations like virus-like particles (VPL) made in a baculovirus insect cell expression system, gene therapy vectors, and even traditional vaccine preparations. Recent projects have included a dual VLP vaccine, recombinant Dengue and RSV vaccines, and a live oral rotavirus vaccine.
As CMOs respond to government and financial pressures by implementing quality and efficiency measures, they see benefits in the bottom line and other fronts. Novel conjugated biologics, though challenging to manufacture, provide yet another opportunity for niche players that can develop an expertise in this new arena. Expanded facility and service offerings are a response to customer demand and will drive revenue growth.
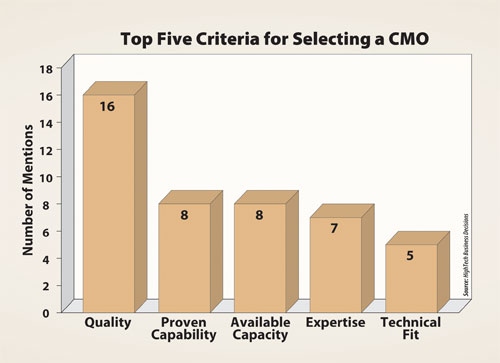
Quality is the top-ranked criteria for CMO selection, according to HighTech Business Decisions’ “Biopharmaceutical Contract Manufacturing Report: Expanding Markets, New Capacities and Improved Performance.”
William Downey ([email protected]) is president, Cindy Liu is managing director, and Jennifer Hartigan is senior scientific director at HighTech Business Decisions.







