September 1, 2009 (Vol. 29, No. 15)
Larger Pool of Potential Research Participants Could Accelerate New Drug Development
Globalization is not new to the world of biomedical and clinical research. For years in the quest for efficiency, biomarker and clinical lab work as well as data management have been moving into developing countries like India. These days the entire clinical research enterprise is a global endeavor.
The rise in international trials, in tandem with concerns that have been expressed in some circles about global clinical research and generally declining participation rates among people in developed countries, led the Association of Clinical Research Organizations (ACRO) to examine the topic in detail.
The most significant finding from our research is that globalization is not driven primarily by cost or regulatory factors but rather by a desire to speed drug development by accessing a larger pool of research participants.
In practical terms, this means getting new medications to market faster. For instance, to fully enroll patients for all current Phase III cancer clinical trials would take 5.8 years if the studies were restricted to the United States. By accessing a global pool of cancer patients, this enrollment time is slashed to 1.9 years.
Critics of globalization ignore these facts and instead tend to focus on incendiary issues like informed consent, developing countries’ regulatory regimes, and research quality. Our research, however, showed that ethical and safety standards, regulatory oversight, and research quality are comparable worldwide, whether a trial is conducted in India or Indiana.
So how prevalent is globalization in clinical research, really? By some estimates, drug companies do as much as one-third of their testing outside of the U.S., Canada, and Western Europe. This is not true, according to the Pharmaceutical Research and Manufacturers of America, which reports that its members in 2007 spent only 3% of their research and development dollars in developing countries.
Frost & Sullivan came to similar conclusions in a recent report, which estimated that in 2008 researchers spent about 49% of research and development money in North America; 37% went to Western Europe; Asia, which includes both industrialized and less-developed countries, got 13.5%; which left less than half a percent for the rest of the world.
International research brings many benefits to the host country including increased spending, access to advanced equipment, highly trained researchers, and high-paying jobs.
Globalization, as we know, is inevitable and inexorable. But in drug testing, we can make sure it is as safe and ethical as possible. We believe that the U.S. FDA should conduct inspections of testing abroad and that the industry should work harder at convincing countries of the importance of a good, ethical infrastructure for drug research.
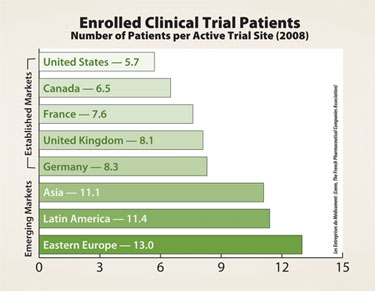
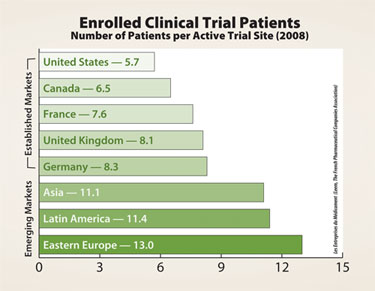
Number of patients per active trial site
Douglas Peddicord, Ph.D. (doug.peddicord @acrohealth.org), is executive director of ACRO. This article is excerpted from “The Case for Globalization: Ethical and Business Considerations in Clinical Research”. The full report is available at www.acrohealth.org/globalization.